Getting a patient diagnosed and prescribed is hard. Keeping them on therapy long enough to see real benefit? That's often even harder — and the stakes couldn't be higher.
Most medications don't deliver their full effect after a single fill. Antidepressants require weeks of consistent use before therapeutic stabilization sets in. Biologics for chronic skin conditions need time to modulate the immune response. Long-term maintenance therapies only work if patients actually maintain them. When patients drop off early, they don't just generate less revenue for the brand — they frequently don't get better.
For pharma and life sciences brands, the gap between prescription and persistence is therefore one of the most costly — and underaddressed — challenges in the business. Patients discontinue therapy for a wide range of reasons: they forget refills, lose touch with their specialist, or simply don't feel the urgency to continue a medication when symptoms aren't immediately visible. The result is a cycle of drop-off that undermines both patient outcomes and brand performance.
Nimble, the platform modernizing independent and regional pharmacy operations, has developed a data-driven approach to solving this problem — and the results across multiple therapeutic areas are compelling.
The Core Idea: Meeting Patients at the Moment That Matters
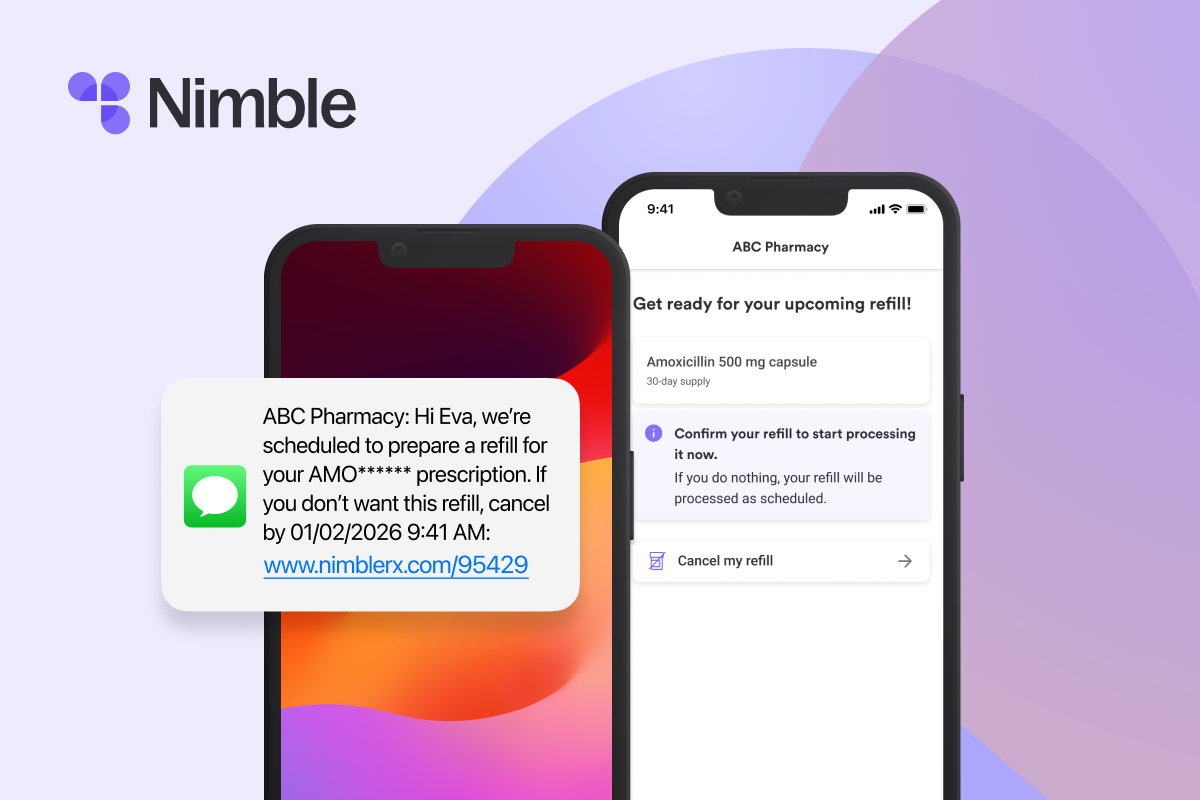
Nimble's patient engagement programs work through the pharmacy layer — a touchpoint that's often overlooked in DTC and patient support strategies but sits at the critical junction between prescription and actual medication use. By leveraging precise digital outreach, Nimble can identify targeted patient populations and deliver timely, relevant interventions that keep them connected to their care plans.
The approach isn't one-size-fits-all. Nimble tailors its programs to the specific clinical and behavioral challenges of each therapy area — whether that's addressing the high discontinuation rates common in mental health, re-engaging patients who've drifted away from specialist care, or sustaining adherence for long-term maintenance medications.
What the Data Shows
Nimble recently published a new case study documenting outcomes across three distinct therapeutic programs: a mental health indication, a dermatology program targeting chronic skin conditions, and a women's health maintenance therapy.
Across all three, the results point in the same direction: patients who receive Nimble's digital support are meaningfully more likely to stay on therapy, refill their prescriptions, and remain engaged with their care providers.
A few highlights from the data:
- Adherence improvements were consistent and significant across all three programs — not marginal gains, but the kind of lift that moves the needle on persistence curves and lifetime patient value.
- Specialist engagement increased substantially in the dermatology program, with targeted outreach driving patients back to their physicians at rates well above the control group.
- New-to-brand starts surged when Nimble identified and activated treatment-naive patients who had previously been managing their condition on inadequate therapies.
- Long-term adherence continued to climb month over month in the women's health program, with dispensed quantities also increasing — a strong indicator that patients weren't just refilling, but actually taking their medication.
Why This Matters for Pharma Brand Teams
For brand managers and patient support leads, the commercial implications of improved persistence are straightforward: more refills, longer treatment duration, and prescriptions that convert into realized, ongoing therapy.
But the most important story here may be the patient outcome one. Medications only work when patients take them — and for most chronic conditions, the clinical benefit compounds over time. A patient with atopic dermatitis who stays on a biologic long enough experiences sustained skin clearance. A depression patient who remains on their antidepressant through the critical stabilization window is far more likely to achieve remission than one who discontinues after a few weeks. A woman who consistently fills her maintenance therapy reduces her long-term health risk in ways a single fill never could. Persistence isn't just a commercial metric — it's a proxy for whether the treatment actually worked. Nimble's programs address both dimensions simultaneously, turning better adherence into better outcomes for patients and better performance for brands.
The case study also speaks to the value of the pharmacy network as a patient engagement channel. Independent and regional pharmacies serve millions of patients who may have less access to the robust support infrastructure that surrounds large health systems. Nimble's technology brings a level of proactive, data-driven outreach to those patients that was previously difficult to achieve at scale.
Read the Full Case Study
The full case study includes detailed program data, outcome charts, and a breakdown of results by therapeutic area — giving brand and marketing teams a clear picture of what Nimble's interventions look like in practice and the kind of impact they can deliver.
To learn more about Nimble's patient engagement programs, visit nimblerx.com.